Thursday, January 5, 2012
Harms of Epidural Steroid Injections Examined
A surge in epidural steroid injections to alleviate back and neck pain in the United States is bringing with it an increase in severe and unexpected complications, including paralysis and death, according to a report in Bloomberg News [here]. Nearly 9-million Americans received these interventional procedures during 2010 alone, and notices of serious adverse effects have prompted the U.S. Food and Drug Administration, in consultation with an advisory group, to review the safety of steroid injections into the epidural space near the spinal cord.
The FDA review comes during a boom in epidural steroid injections, which take minutes to administer and reap profitable reimbursements from Medicare and private insurers, the Bloomberg report observes. One study, by Laxmaiah Manchikanti, MD, chairman of the American Society of Interventional Pain Physicians (ASIPP), found the number of such injections among Medicare patients increased 159% between 2000 and 2010. Epidural injections are one of many interventional procedures — including implants of spinal cord stimulators — on which Americans spent $23 billion in 2011, up by 231% from 2002, the Bloomberg report notes.
The rise in epidural injections is being driven by two factors, the Bloomberg article claims:
- an aging population prone to back and neck pain, and
- generous reimbursements for interventional pain management procedures.
According to James Rathmell, MD — chief of pain medicine at Massachusetts General Hospital in Boston who alerted the FDA to cases of such injections causing harsh complications and is a member of the agency’s advisory group — “The problem with interventional pain is the majority of treatment is medical management. If you pay people to do stuff, they will do more stuff.”
David Armstrong, who authored the Bloomberg article, found that Medicare pays about $200 for a typical epidural steroid injection if given in a doctor’s office, roughly $400 if done at a surgery center, and about $600 if performed at a hospital. Some private insurers reimburse as much as 150% of Medicare rates. Meanwhile, the cost of the equipment, supplies, and staffing needed for a typical injection can be as low as $120.
The drugs used for epidural injection are anti-inflammatory corticosteroids, which are popular for easing pain in knees, hips, shoulders, and other parts of the body, in addition to the neck and back. The FDA review of epidural injections is being conducted by the agency’s “Safe Use Initiative,” a unit formed in 2009 to reduce “preventable harm” from medications. The focus of their investigation will be on steroid injections via the transforaminal approach, which brings a needle within millimeters of critical arteries feeding the spinal cord (also see explanation below). About half of the 8.9 million epidural steroid injections in the U.S. last year were administered using that approach, according to Manchikanti.
Another area of concern noted in the Bloomberg report is the use of particulate steroids. This form of the drug is slow to dissolve and may create blockages that trigger strokes if accidentally injected into arteries. Labeling of these agents specifies that they are not indicated for epidural use and that serious adverse events, including death, may occur.
At one time, serious adverse events with epidural steroid injections were thought to be rare or, at worst, relatively minor, such as headaches. “Then researchers and malpractice insurance companies began learning of cases of people becoming paralyzed and even dying after receiving the shots,” the Bloomberg report claims. A survey of physicians, reported in the journal Spine in 2007, revealed 78 cases where patients who received cervical injections suffered serious injuries; there were 13 deaths.
Additionally, an analysis by Rathmell and colleagues of malpractice claims between 2005 and 2008 identified 31 cases in which patients receiving cervical injections reported spinal cord injuries, and 8 who suffered strokes. In the analysis, Rathmell et al. called the cases “alarming” and said there is an “extreme dearth of evidence” about the safety of the injections.
Richard Rosenquist, MD, chairman of the pain management department at the Cleveland Clinic said in the Bloomberg article that part of the danger is due to the fact that almost any physician can give someone a spinal injection. “The unsuspecting public has no idea someone might have gone to a weekend course and on Monday morning is testing out their brand new skill on you. It’s horrible,” he stated.
Furthermore, the Bloomberg article observes that “Medicare patients receive transforaminal epidurals in a physician’s office more than any other setting,” according to a 2010 government audit. A third of those injections did not meet Medicare requirements — 13% because they were not deemed medically necessary and the rest because providers submitted bills lacking documentation as to why the injections were needed or improperly coded procedures. In some cases, according to the audit, multiple injections were given at close time intervals with no evidence that they were relieving pain.
How many injections are appropriate? According to the Bloomberg report, the North American Spine Society suggests a maximum of 4 injections within 6 months for cervical epidurals, but adds that setting absolute limits on the number could inappropriately limit some patients from receiving necessary care. Guidelines for pain management from the American Society of Anesthesiologists make no mention of how many injections are appropriate. The Mayo Clinic in Rochester, Minnesota, tells patients that injections are usually limited to a few annually because steroids can weaken spinal bones and nearby muscles and upset natural hormone balance, leading to potentially serious medical conditions. As might be expected, risks of side effects or serious adverse events might increase with a greater frequency of steroid injections.
COMMENTARY: In an UPDATE last September [here] we discussed an essay by Gerald M. Aronoff, MD, DABPM, claiming that the pain management field has been burdened by escalating interventional procedures that may be driven more by economics than better patient care. And, as a result of the ongoing turf wars between interventionists and medical practitioners, patients with pain are in danger of becoming collateral damage.
According to the Bloomberg News report described above, epidural steroid injections for neck and back pain are a burgeoning business. The injections are typically used to treat spinal stenosis, spondylolysis, herniated disc, degenerative discs, or sciatica and other spinal nerve entrapments. However, because the anti-inflammatory effects of the steroid agents, which are usually mixed with a local anesthetic, are short-term, the injections provide only temporary relief of pain, if any at all, and repeat procedures with their potential risks may be necessary.
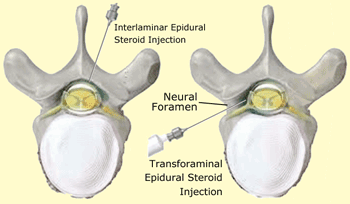
Two approaches to the epidural space — inside the bony spinal canal but outside the dura mater enveloping the spinal cord — are commonly mentioned in the literature, interlaminar and transforaminal [see drawing]:
- The interlaminar approach involves insertion of an injection needle into the epidural space midway between two adjacent vertebrae.
- The transforaminal approach entails insertion of a needle into the intervertebral neural foramen (or, hole) where the nerve root exits the spine.
A third variation, used less often, is the caudal epidural steroid injection [not shown in the drawing] in which the needle is placed through a small opening at the base of the spine just above the tailbone.
In an excellent review of the literature comparing interlaminar versus transforaminal injections, Christopher Huston [2009] observes that wide variations in success rates with the interlaminar approach have fostered increased interest in transforaminal epidural injections, since these deliver targeted solution closer to the nerves generating pain. Huston claims that this approach is more effective than interlaminar or caudal injections and fluoroscopy-guided transforaminal injections have “emerged as the preferred approach to deliver steroids to the epidural space.”
However, because this approach also sends the needle so near to vital arteries, other practitioners believe the dangers of transforaminal approaches are too high. In the Bloomberg article, Manchikanti states that the transforaminal approach for cervical injections, in particular, has no role when considering the risks and that the organizatin he leads, ASIPP, has stopped teaching the technique.
Adverse effects of epidural injections have been poorly assessed in much of the research literature and are quite variable. Huston [2009] notes a long list of potential complications, including: dural puncture, nausea and vomiting, vasovagal reaction, facial flushing, fever, nerve root injury, pneumocephalus, epidural hematoma, subdural hematoma, Cushing’s syndrome, paresthesias, hypotension, non-specific headache, transient amnesia, increased pain, infection, respiratory insufficiency, transient blindness, epidural abscess, paralysis, spinal cord injury, cerebellar infarction, stroke, and death. He also notes that rates of the more transient or minor complications have variably ranged up to about 23%; although, there can be delayed reactions and complications days or weeks after the procedures that often go unreported in research studies. Incidence rates of catastrophic neurologic complications and death, while believed to be relatively rare, are unknown for certain.
Similarly, the degree of pain relief and its duration afforded by epidural injections also are highly variable, and appear to depend on numerous factors, including: the etiology of pain and its spinal location, specific solutions/mixtures and dosages used, administration technique, and the experience and skills of the individual practitioner. Large-scale, randomized, controlled clinical trials have been scarce, and the results of observational studies have been mixed, with both favorable and unfavorable outcomes.
Despite the concerns, Huston [2009] notes in his review that 40% to 85% of patients have successful long-term (more than 3 months) pain relief from transforaminal epidural steroid injections. This procedure also can be helpful to prognosticate potential success of surgery, if needed; patients who had 70% to 80% relief from transforaminal steroid injections were shown to experience greater than 95% success in achieving an average of 90% pain relief following surgery.
Often, however, outcomes comparing injection modalities in higher quality clinical trials are equivocal. Two very recently reported clinical trials exemplify this:
- In a prospective, blinded, randomized trial of interlaminar versus transforaminal epidural steroids for subacute low back pain with radiculopathy [Gharibo et al. 2011], the transforaminal approach was superior in lowering pain ratings. However, on all other endpoint measures — disability, function, depression, and opioid use — there were no difference between the two techniques; both approaches were considered to be efficacious.
- In a randomized, placebo-controlled trial of epidural steroid injections for chronic radiculopathy [Iversen et al. 2011], patients were injected caudally with either saline solution (placebo control) or saline + corticosteroid (active therapy), or subcutaneous saline (sham control). While there were small improvements in all 3 treatment conditions these did not reach statistical significance or differ between groups.
Each study had limitations and enrolled only carefully selected patients, which might have biased outcomes. The studies also were of insufficient size to assess the potential for adverse effects, which is a typical deficiency of research involving interventional pain management techniques.
Obviously, this is a topic worthy of much more thorough investigation. However, from the Bloomberg News report and the several other papers examined above, it seems evident that epidural steroid injections represent a growing area of interventional pain medicine that is laden with some concerns and controversies. While the procedures are very commonly performed for the symptomatic relief of a variety of pain conditions, they are temporary measures with many potential adverse effects, some more common and less serious than others. However, the need for repeated procedures to achieve ongoing pain relief also might increase the probability of adverse events occurring.
Currently, there appear to be some disagreements among experts, and conflicting clinical evidence, as to which anatomical approaches for injection are best, and there are a number of significant factors that can influence successful versus potentially harmful outcomes. Clearly, therapeutic decisions regarding epidural injection interventions should be pursued cautiously and with full consideration of benefits versus risks.
REFERENCES:
> Gharibo CG, Varlotta G:, Rhame EE, et al. Interlaminar versus transforaminal epidural steroids for the treatment of subacute lumbar radicular pain: a randomized, blinded, prospective outcome study. Pain Physician. 2011(Dec);14(6):499-511 [access article here].
> Huston CW. Cervical epidural steroid injections in the management of cervical radiculitis: interlaminar versus transforaminal. A review. Curr Rev Musculoskelet Med. 2009;2(1):30-42 [access article here].
> Iversen T, Solberg TK, Romner B, et al. Effect of caudal epidural steroid or saline injection in chronic lumbar radiculopathy: multicentre, blinded, randomized controlled trial. BMJ. 2011;343:d5278 [article here].